T-E Pharma has officially launched its pre-initial public offering (pre-IPO) fundraising program. The proceeds will be used to accelerate the development of its novel therapeutic pipeline, expand its global strategic presence, and strengthen its capital structure. The company is also actively pursuing international partnership opportunities, with the goal of listing on the Taiwan Emerging Stock Market in Q4 2026 and ultimately becoming a globally competitive innovative drug development company.
Immunwork Inc., the first subsidiary of T-E Pharma, was founded in 2014 by internationally renowned drug development expert Dr. Tse-Wen Chang. The company is dedicated to the research, design, and development of innovative therapeutics. During its early years, Dr. Chang served as the sole investor for approximately six years, a period in which the company established key platform technologies, validated multiple drug candidates, and built a robust intellectual property portfolio. On this foundation, the company completed its first Series A financing round of approximately US$24 million in 2021, enabling multiple candidates to advance into pre-clinical development.
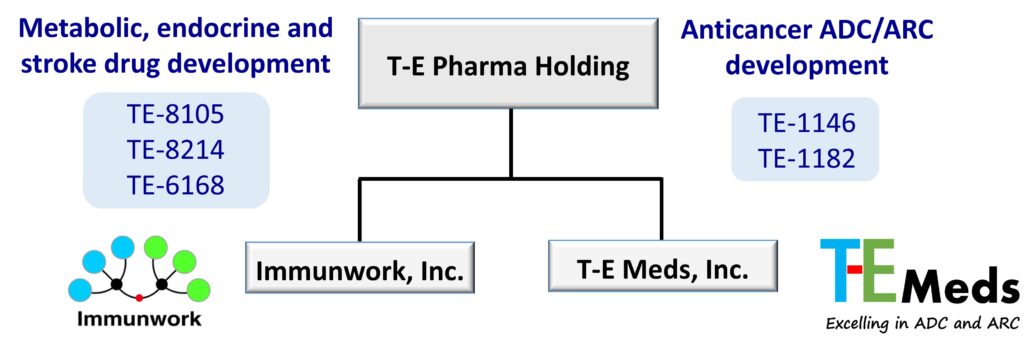
To further enhance R&D efficiency and expand its pipeline, T-E Pharma established its second subsidiary, T-E Meds Inc., in 2022. The company subsequently completed two rounds of capital increases totaling approximately US$26 million, with a focus on the development of antibody-drug conjugates (ADCs) and antibody-radionuclide conjugates (ARCs). As the group continued to scale, T-E Pharma completed a corporate restructuring in 2025, positioning itself as a holding and capital management platform with full ownership of both Immunwork Inc. and T-E Meds Inc. This structure enables clear operational segmentation and is designed to support the company’s international capital market ambitions.
Modular Platform Technology Driving Innovative Drug Design
As an R&D-driven biotechnology company, T-E Pharma’s competitive advantage lies in its proprietary “T-E pharmaceutical” design concept, integrated with its internally developed “Multi-arm linker” core platform. This technology has given rise to three modular sub-platforms: the “Fatty Acid Bundle” platform, the “Cytotoxic Drug Bundle” platform, and the “Chelator Bundle” platform. Together, these platforms enable flexible molecular engineering across diverse therapeutic modalities with improved efficacy and safety profiles, demonstrating strong scalability and broad application potential.
Dual-Engine Strategy Targeting the Metabolic and Oncology Markets
To enhance R&D efficiency and mitigate development risk, T-E Pharma has adopted a dual-engine operational model in which its subsidiaries focus on distinct therapeutic areas.
Immunwork, Inc.: Metabolic, Endocrine, and Stroke Therapeutics
Immunwork leverages the “Fatty Acid Bundle” platform to develop long-acting therapeutics targeting large metabolic and endocrine disease markets, including diabetes, obesity, non-alcoholic fatty liver disease, acromegaly, and neuroendocrine tumors. The company is also advancing a novel therapeutic candidate that addresses the significant unmet medical need in ischemic stroke, an area with strong clinical potential.
T-E Meds, Inc.: Anticancer ADC/ARC Development
T-E Meds specializes in high-barrier anticancer drug development. Through its “Cytotoxic Drug Bundle” and “Chelator Bundle” platforms, the company is advancing a pipeline of ADCs and ARCs, aiming to provide more precise and effective treatment options for patients with advanced and difficult-to-treat cancers.
(Appendix: T-E Pharma Organizational Structure)
Clinical Progress Validates Platform Potential
The company’s two lead clinical candidates, TE-8105 and TE-8214, are currently in clinical trials. Preliminary results demonstrate favorable safety and tolerability profiles, and dose-escalation and efficacy evaluation studies are progressing steadily. As clinical data continue to accumulate, the company is actively laying the groundwork for future global commercialization and strategic partnership opportunities.
Advancing Toward Capital Markets and Global Biopharma Leadership
T-E Pharma stated that proceeds from the current fundraising round will be allocated to advancing clinical trials, expanding the pipeline of high-potential assets, strengthening international strategic collaborations, and supporting pre-IPO operational and regulatory preparations. With its technology platforms and product pipeline continuing to mature, the company is actively developing its IPO roadmap and aims to list on the Taiwan Emerging Stock Market in Q4 2026.
Looking forward, T-E Pharma aspires to leverage capital market support to evolve into a world-class innovative drug development company—one that originates from Taiwan and delivers meaningful therapeutic benefits to patients worldwide.
(Appendix: Overview of T-E Pharma Pipeline and Latest Development Progress)
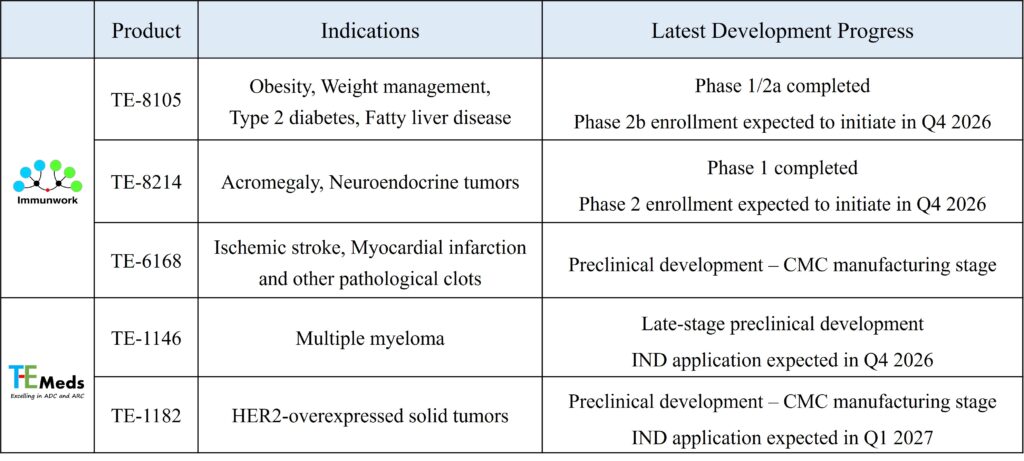
Note 1: TE-8105 and TE-8214 are classified as novel drugs under the 505(b)(1) regulatory pathway. TE-6168, TE-1146, and TE-1182 also feature entirely new chemical entities.
Note 2: TE-8105, TE-6168, and TE-1146 were designated as priority drug development cases by the Center for Drug Evaluation (CDE) in 2019, 2026, and 2025, respectively.
